Biden organization officials are set to declare Tuesday a pristine strategy to isolate monkeypox immunization dosages in order to immunize as much as 5 events as numerous people contrary to the infection, with regards to officials with direct information of the arrangement.
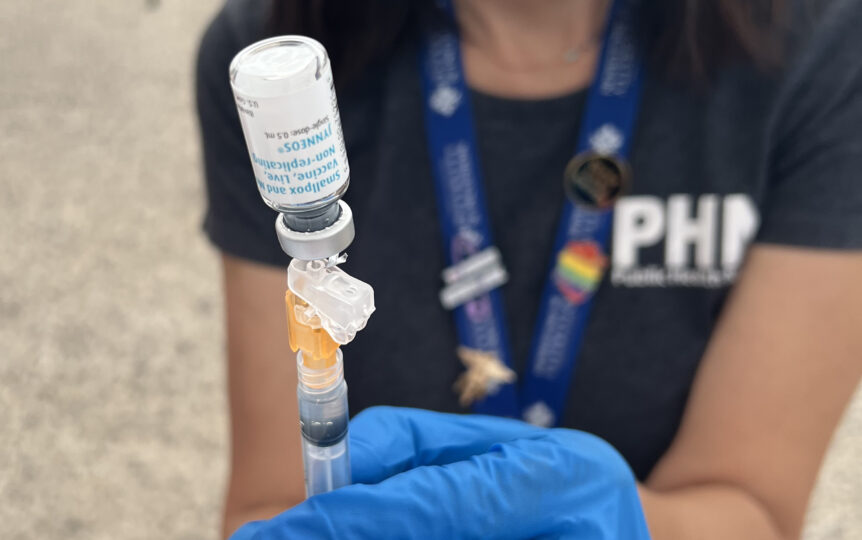
The method, first depicted freely by Meals and Drug Administration Commissioner Robert Califf last week, would empower the Biden organization to extend its restricted stock of monkeypox antibodies by adjusting how these photos are regulated. Very than infuse dosages of Jynneos subcutaneously, a traditional methodology of conveying immunizations into the greasy tissue under the pores and skin, the portions would as an option be infused under the most elevated layer of the pores and skin. This strategy, alluded to as an intradermal infusion, utilizes a more slender needle and less immunization, but brings about a little air pocket framing on the floor of the pores and skin that might scar.
The adjustment of infusion method would amplify the resistant reaction produced by the antibody and grant U.S. officials to exclusively control one-fifth of the novel portion, Califf trained columnists last Thursday, focusing on that the strategy wouldn’t think twice about or viability.
“It really implies, chiefly, staying the needle all through the pores and skin and making a tad pocket there into which the immunization goes,” Califf referenced, assessing it to tests for tuberculosis and various infusions did by medical services staff. “That is quite very phenomenal.”
Anyway the conscious change in immunization dosing could be an enormous scope, constant examination as officials competition to fight off a monkeypox flare-up that has polluted more noteworthy than 8,900 people in america. In the event that productive, the pristine immunization plan would empower the Biden organization to redesign lots of 1000’s of current portions of Jynneos — the one FDA-supported antibody for monkeypox — into various million possible photos. However U.S. officials last month got for all intents and purposes 800,000 further portions of Jynneos, request has quickly outperformed supply and additional dosages won’t be expected for a really long time.
The change simply isn’t with out danger. People who get the pared-down model of the Jynneos immunization, which is implied as a two-portion schedule, could end up requiring further photos in the event that the spic and span immunization procedure brings about a deficient phase of security contrary to the infection. Local officials could likewise fight to deal with the fresh out of the box new method, which could require further training and gives like new needles to transport the photos sufficiently.
Battle to shield gay men from monkeypox uncovered imbalances in local area
“This isn’t a ‘snap your fingers and it’s finished,’ ” referenced a public prosperity talented who was informed by the organization on its forthcoming arrangement anyway was not endorsed to comment. “There’s loads of intricacy to carrying this out appropriate, numerous inoculation sites gained’t be totally ready, and it’s not obvious that [the U.S. Division of Health and Human Services] has finished each easily overlooked detail it should to help get local arrangements set up.”
Michael Osterholm, overseer of the Middle for Infectious Illness Analysis and Coverage on the College of Minnesota, referenced he had contemplations concerning the insusceptible security presented by the method, outstandingly for people with prosperity circumstances like HIV. “We don’t yet have the data to comprehend how effectively an intradermal-controlled immunization will guard immunocompromised people,” he referenced.
HHS didn’t immediately answer to a solicitation for comment. The FDA referenced Monday that the organization had no data to share.
Also Read:Billie Shepherd Shows Off Her Blowing Baby Bump
Organization officials toiled through the end of the week on the operations of the pristine procedure, bringing on earlier investigation into parting antibody dosages, which is at times called “portion saving.” Infectious-illness gifted Anthony S. Fauci and different senior organization officials have checked on that information and help the conscious change, with regards to two officials who talked on the circumstance of namelessness because of they weren’t endorsed to comment.
The government specialists ought to regardless make various strides sooner than the immunization change is lawfully allowable. However Well being and Human Companies Secretary Xavier Becerra last Thursday pronounced monkeypox a public prosperity crisis, he would need to trouble a second statement that might empower for “crisis use” of the predominant monkeypox immunizations and different clinical countermeasures, such on the grounds that the arrangement to change how the photos are directed.
The Facilities for Illness Management and Prevention can be locked in on steerage for medical services staff on how one can direct the shiny new immunization strategy, with regards to two unique people with information of the purposeful declaration who talked on the circumstance of obscurity because of they weren’t supported to comment.
U.S. rehashes early Coronavirus botches in monkeypox reaction, specialists say
Outside experts flagged their help for the idea. Daniel McQuillen, leader of the Infectious Illnesses Society of America, referenced in a public statement that his gathering “concurs with the method into thought by FDA.”